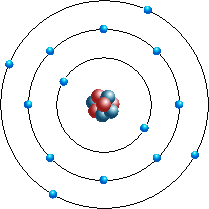
Silicon
Silicon is a chemical elements with symbol Si and atomic number 14. It is a hard and brittle crystalline solid with a blue-gray metallic lustre; and it is a tetravalent metalloid and semiconductor. It is a member of group 14 in the periodic table : carbon is above it, and germanium, tin, and lead are below it.
|
Stability....As noted above, benzene is thermodynamically unstable compared with carbon and hydrogen, but it is chemically stable under normal laboratory conditions, even when mixed with some reactive compounds such as bromine. Compounds or mixtures that are chemically unstable are often called labile. |
Elements can be classified based on their physical states ( States of Matter) e.g. gas, solid or liquid. This element is a solid. Silicon is classified as a ¨Metalloid¨ element and is located in Groups 13, 14, 15, 16 and 17 of the Periodic Table. |